The Puna plateau sits at an elevation of 4,000m, stretches for 1800 km. along the Central Andes and attains a width of 350–400 km. The Puna covers a portion of Argentina, Chile and Bolivia and hosts an estimated 70%–80% of global lithium brine reserves.
The evaporate mineral deposits on the plateau, which may contain potash, lithium and boron, are formed by intense evaporation under hot, dry and windy conditions in an endorheic basin—endorheic basins are closed drainage basins that retain water and allow no outflow—precipitation and inflow water from the surrounding mountains only leaves the system by evaporation and seepage. The surface of such a basin is typically occupied by a salt lake or saltpan. Most of these salt lakes, called salars, contain brines that are capable of providing more than one potentially economic product.
This Puna Plateau area of the Andean mountains—where the borders of Argentina, Bolivia and Chile meet and bounded by the Salar de Atacama, the Salar de Uyuni and the Salar de Hombre Muerto—is often referred to as the Lithium Triangle and the three countries mentioned are the Lithium ABC's.
Brine "Mining"
The salt rich brines are pumped from beneath the crust that's on the salar and fed into a series of large, shallow ponds. Initial 200 to +1,000 parts per million (ppm) lithium brine solution is concentrated by solar evaporation and wind up to 6,000-ppm lithium after 18–24 months.
The extraction process is low cost/high margin and battery grade lithium carbonate can be extracted.
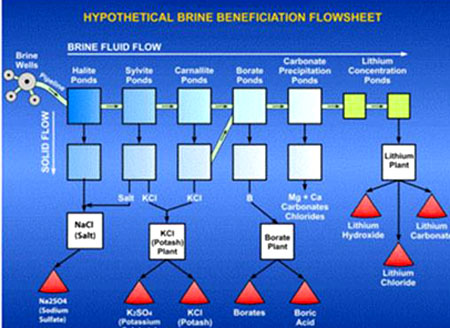
The above diagram was designed to show that several commercial products can be recovered from typical brine and that the recovery takes place in a series of steps over the entire evaporation process. Note that the final product in each step may require processing in a specialized plant. Also please note that the actual sequence of process steps may vary from brine to brine, and as such, the process steps shown above may not be in the correct order for any specific brine.
The key factors that determine the quality, economics and attractiveness of brines are:
- Potassium content
- Lithium content
- Presence of contaminants (i.e., magnesium [Mg])
- Porosity
- Net evaporation rate
- Recoverable byproducts
- Infrastructure—or lack thereof
- Country risk
- 100% control over production
- Low capex, low production costs, high-margin products
A common industry axiom says that the ratio of Mg to Li in brines must be below the range of 9:1 or 10:1 to be economical. This is because the Mg has to be removed by adding slaked lime to the brine—the slaked lime reacts with the magnesium salts and removes them from the water.
The porosity of a rock is expressed as a percentage, and refers to that portion of the rock that is void space—rock that is composed of perfectly round and equal sized grains will have a porosity of 45%. Fluids and gases will be found in the void spaces within the rock.
Ten million cubic meters of brine-bearing rock with a porosity of 10% will contain 1 million cubic meters of brine fluid. A cubic meter is equivalent to a kiloliter.
By oil and gas standards, a porosity of 10% is quite low, but brines are less viscous than hydrocarbon fluids and will flow more easily through rocks with lower porosity and permeability characteristics.
A major factor affecting capital costs is the net evaporation rate; this determines the area of the evaporation ponds necessary to increase the grade of the plant feed. These evaporation ponds can be a major capital cost. The Salar de Atacama has higher evaporation rates (3200M pan evaporation rate per year (py) and <15M py of precipitation) than other salt plains in the world and evaporation takes place all year long.
Contributing to efficient solar evaporation and concentration of the Puna Plateau brines are:
- Low rainfall
- Low humidity
- High winds
- High elevations
- Warm days
A company should have 100% control over the production rate from their salar. It's possible an aquifer can become diluted—over producing can impact the brine's salt concentrations and chemical compositions—or depleted by too many wells sucking up more brine than should be produced.
If two or more companies have straws (wells) into the same salar legal battles might result over the sharing of the resources.
Potash Is Fuel for Food
Potash is used as a major agricultural component in 150 countries, but the largest potash importers are China, India, the U.S. and Brazil.
Potassium sulfate is commonly used in fertilizers, providing both potassium and sulfur. Potash is the common name for potassium chloride.
The basic fundamentals of the global potash market are hard to ignore:
- The increasing global population is expected to reach +9 billion people by 2050. The United Nations Food and Agriculture Organization (FAO) reported they think that the total world demand for agricultural products will be 60% higher in 2030 than it is today.
- Increasing incomes in developing countries will lead to more people being able to afford protein rich diets—a Western-style diet heavy in meat—which means more grain consumption.
- Decreasing arable land—arable land is being lost at the rate of about 40,000 square miles per year. Land is being used for production of bio-fuels, topsoil is eroded away by wind and water and the agriculture land base is being paved over as we become more and more urbanized. Farmers need to produce more food on less land.There is only one way this can be done and that's with an increase in the use of fertilizer.
The current potash market is estimated at 50 million tons (Mt.) annually and is projected to grow at a compounded annual rate of 3%–4%. Potash is a crucial element in fertilizer and has no commercial substitute.
Lithium
The world's future energy course is being charted today because of the ramifications of peak oil and a need to reduce our carbon footprints.
A whole new industry—a worldwide automotive and industrial lithium-ion battery industry—is being built. As a result of lithium-ion battery demand for hybrid-electric and electric cars the increase in demand for lithium carbonate is expected to increase fourfold by 2017.
Lithium-ion batteries have become the rechargeable battery of choice in cell phones, computers, hybrid-electric cars and electric cars. Chrysler, Dodge, Ford, GM, Mercedes-Benz, Mitsubishi, Nissan, Saturn, Tesla and Toyota have all announced plans to build lithium-ion battery powered cars.
Lithium carbonate is also an important industrial chemical:
- It forms low-melting fluxes with silica and other materials.
- Glasses derived from lithium carbonate are useful in ovenware.
- Cement sets more rapidly when prepared with lithium carbonate, and is useful for tile adhesives.
- When added to aluminum trifluoride, it forms LiF which gives a superior electrolyte for the processing of aluminum.
- Lithium carbonate can be used in a type of carbon dioxide sensor.
Lithium is not traded publicly and is usually distributed in a chemical form, such as lithium carbonate (Li2CO3). Instead, it's sold directly to end users for a negotiated price per ton of Li2CO3.
Production figures are often quoted in lithium carbonate equivalent quantities. By weight, approximately 18.8% of Li2CO3 is lithium. Therefore, 1 kg. lithium is equivalent to 5.3 kg. Li2CO3.
Lithium-ion batteries are quickly becoming the most prevalent type of battery used in everything from laptops and cell phones to hybrid cars and short-term power-storage devices for wind- and solar-generated power.
Sodium Chloride (Rock Salt or Halite)
The principal use for salt is in the chemical manufacturing business—chloralkali and synthetic soda ash producers use salt as their primary raw material.
Salt is used in many applications and almost every industry:
- Cooking
- Manufacturing pulp and paper
- Setting dyes in textiles and fabric
- Producing soaps, detergents and other bath products.
- Major source of industrial chlorine and sodium hydroxide.
Global demand for salt is forecast to grow 2.5% per year to 305 Mt. in 2013.
Solar evaporation is the most popular and most economical method of producing salt. China is the world's largest consumer of salt—other than the dietary needs of 1.3 billion people—there's an enormous chemical manufacturing industry being built in China.
Boron
Boron combines with oxygen and other elements to form boric acid, or inorganic salts called borates. Borates are used for:
- Insulation fiberglass
- Textile fiberglass
- Heat-resistant glass
- Detergents, soaps and personal care products
- Ceramic and enamel frits and glazes
- Ceramic tile bodies
- Agricultural micronutrients
- Wood treatments
- Polymer additives
- Pest-control products
- Boron is an essential component in the manufacture of borosilicate glass used in LCD screens
Boric Acid uses:
- As an antiseptic/anti-bacterial compound
- Insecticide
- Flame retardant
- In nuclear power plants to control the fission rate of uranium*
- As a precursor of other chemical compounds
*Boric acid is used in nuclear power plants to slow down the rate at which fission occurs. Boron is also dissolved into the spent fuel cooling pools containing used fuel rods. Natural boron is 20% boron-10, which can absorb a lot of neutrons. When you add boric acid to the reactor coolant, or the spent fuel rod cooling pools, the probability of fission is reduced.
World production of borates remains mostly concentrated in the U.S. and Turkey. These two countries account for 75% of supply.
Chinese boron, in terms of both quantity and grade, is inadequate to meet domestic demand, so the country is now the largest importer of both natural borates and boric acid.
Conclusion
Potash and agriculture will be one of the top investment themes over the next 20 to 30 years—world population growth and three billion people climbing the protein ladder are elephants in the dining room. Our population has nearly doubled since 1970. We add 80 million people to our global population each year—tonight there will be 220,000 new mouths to feed at the dinner table.
The rechargeable power needs of our modern society have made lithium a serious player in the commodity markets. Lithium makes an excellent battery for use in a wide range of applications—batteries using lithium have been found to have a high energy to weight ratio, can be molded into amazing shapes and have longer lives than conventional batteries. And when used as a rechargeable battery there is no memory effect.
There are significant savings to be had in the pricing of lithium from brines for lithium miners with quality projects close to all necessary infrastructure.
Investing in a macro trend has always been the most dependable way to make money—rising food prices are a macro trend with a long-term time horizon, so is the electrification of our transportation system.
Are both these global macro trends on your radar screen? If not, maybe they should be.
Richard (Rick) Mills
[email protected]
aheadoftheherd.com
If you're interested in learning more about the junior resource, biotech and technology sectors please come and visit us at Ahead of the herd. Site membership, and our AOTH newsletter, are free. No credit card or personal information is required.
Richard is host of Ahead of the herd and invests in the junior resource sector. His articles have been published on over 300 websites, including: The Wall Street Journal, USAToday, National Post, Vancouver Sun, Huffington Post, The Gold/Energy Reports, Calgary Herald, Forbes, SafeHaven, Market Oracle, Stockhouse, Lewrockwell, Uranium Miner, Casey Research, 24hgold, SilverBearCafe, Infomine, Mineweb, 321gold, Kitco, Gold-Eagle, Resource Investor, Mining.com, FNArena, UraniumSeek and Financial Sense.
Legal Notice/Disclaimer: This document is not and should not be construed as an offer to sell or the solicitation of an offer to purchase or subscribe for any investment. Richard Mills has based this document on information obtained from sources he believes to be reliable but which has not been independently verified; Richard Mills makes no guarantee, representation or warranty and accepts no responsibility or liability as to its accuracy or completeness. Expressions of opinion are those of Richard Mills only and are subject to change without notice. Richard Mills assumes no warranty, liability or guarantee for the current relevance, correctness or completeness of any information provided within this Report and will not be held liable for the consequence of reliance upon any opinion or statement contained herein or any omission. Furthermore, I, Richard Mills, assume no liability for any direct or indirect loss or damage or, in particular, for lost profit, which you may incur as a result of the use and existence of the information provided within this Report.
For site advertising rates contact: aheadoftheherd.com
Richard Mills does not own shares of any companies mentioned in this report.





















































